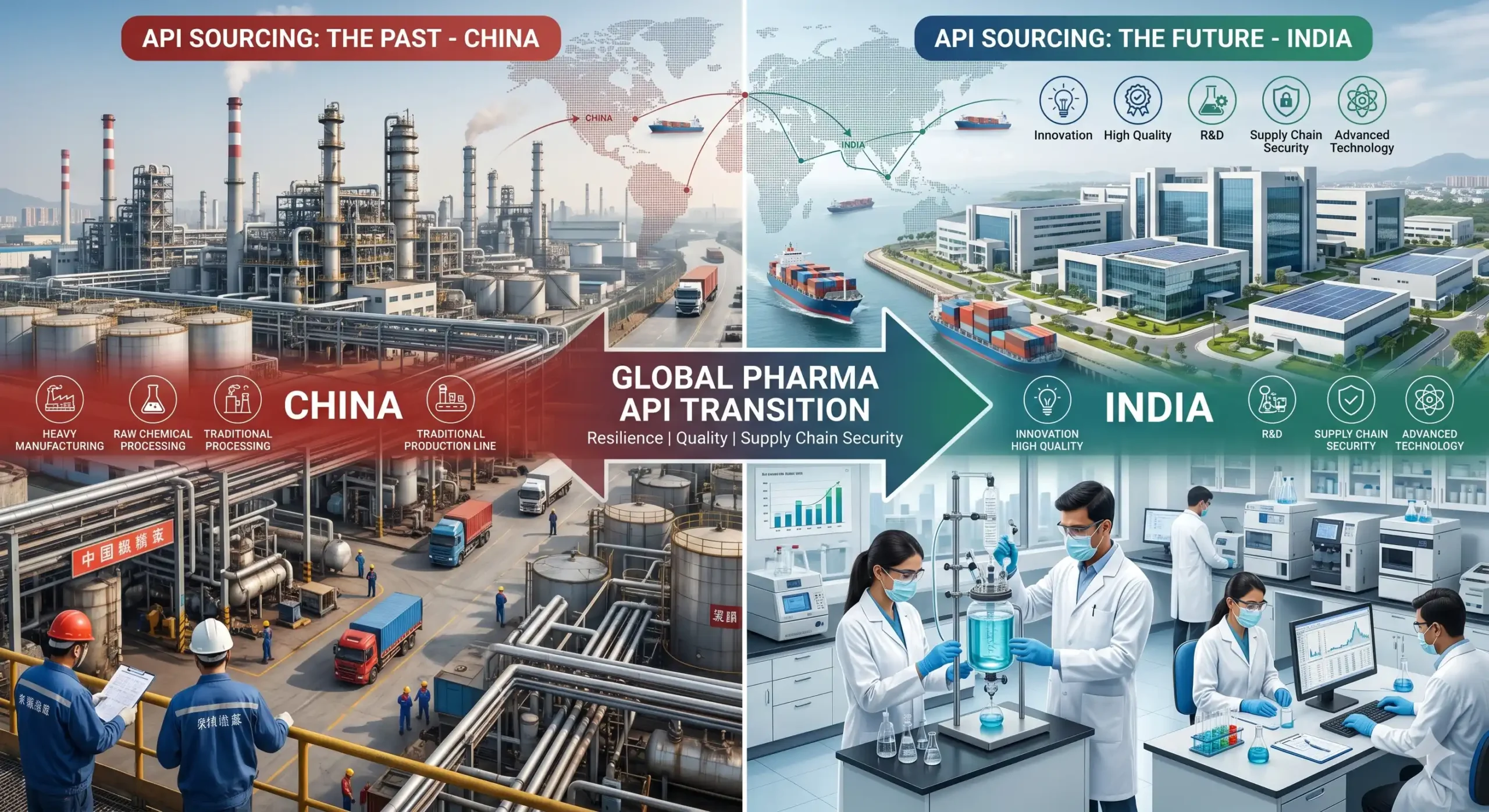
For decades, China’s dominance in Active Pharmaceutical Ingredient (API) manufacturing looked unassailable. Low-cost production and massive scale made it the default choice for global procurement teams. But geopolitical tensions, pandemic-era […]
Uncategorized

Uncategorized
Navigating the 2026 Regulatory Convergence: A Strategic Guide to Schedule M and IP 2026 Compliance
By SEO Support
- March 30, 2026
For pharmaceutical procurement managers and Quality Assurance (QA) directors, 2026 represents the most significant regulatory overhaul in a generation. The era of “informal” manufacturing has ended, replaced by a mandate […]

Blogs - Uncategorized
The 2026 Pharmaceutical Sourcing Masterclass: High-Purity Intermediates, Schedule M Compliance, and the CDMO Shift
By SEO Support
- March 29, 2026
For pharmaceutical procurement managers, global supply chain directors, and technical R&D heads, the operational landscape of March 2026 is defined by a singular, unforgiving mandate: Resilience through Uncompromising Technical Compliance. […]

Blogs - Uncategorized
Pharmaceutical End Products Enabled by Key Heterocyclic Intermediates
By SEO Support
- March 9, 2026
Which pharmaceutical and specialty chemical products rely on heterocyclic intermediates? Many pharmaceutical and specialty chemical products rely on heterocyclic intermediates such as pyrimidines, isatin derivatives, and quinoline compounds, which enable […]
Recent Posts
- Why India is Emerging as a Global Hub for Specialty Chemical Manufacturers and Exporters
- How to Choose a Reliable Pharmaceutical Intermediates Manufacturer in India
- The Role of 7-Chloroquinaldine as a Pharmaceutical Intermediate in Respiratory Medicine
- Setting New Purity Benchmarks for 5-Bromo-2-Chloropyrimidine Achieved by Advance Pharma Chem
- 7-Chloroquinaldine: Sourcing Guide for Bulk Buyers – Purity Standards, CAS Verification, & more
Recent Comments
No comments to show.